2On February 18, 2026, Associate Professor Wang Lu and her coworkers from our institute published their latest research findings online in the internationally renowned journal Angewandte Chemie International Edition (Q1, Impact Factor: 16.9). The paper, titled "Multiscale Engineering of PEO Electrolytes for High-Voltage and Ultrastable Solid-State Lithium Batteries With Exceptional Room-Temperature Performance," represents a significant breakthrough. Graduate student Liu Xuefan and Professor Zhang Bowen are the co-first authors. Corresponding authors include Associate Professor Wang Lu, Associate Professor Kong Linglong from the College of Forestry, and Professor Zhang Shanqing from Guangdong University of Technology. Shandong Agricultural University is the primary affiliation for this paper.。

Solid-state batteries, offering inherent safety and high energy density, are widely recognized as a core direction for next-generation electrochemical energy storage. Among various electrolytes, poly(ethylene oxide) (PEO)-based solid electrolytes are the most extensively studied polymer system, leveraging the high donor number (DN) and chain flexibility of ethylene oxide (EO) units for superior lithium salt dissolution and ion transport. However, PEO electrolytes have long faced three major challenges: low ionic conductivity at room temperature, poor compatibility with high-voltage cathodes (prone to oxidative decomposition above 4 V), and insufficient cycling stability. Furthermore, current research often relies on adding liquid plasticizers or gelation to enhance room-temperature performance, which compromises the inherent safety of solid-state batteries.
To address these challenges, the research team proposed a liquid-plasticizer-free and multiscale engineering strategy. By synergistically optimizing the macroscopic structure, mesoscopic segmental motion, and microscopic solvation configuration of PEO electrolytes, they achieved ultra-long lifespan cycling for lithium metal batteries under high voltage and room temperature conditions without introducing any liquid components. This provides a proof-of-concept for designing high-performance PEO electrolytes. Specifically, a PEG-PDMAEMAH+·NO3? (abbreviated as PD) copolymer is designed and synthesized and introduced it into a high molecular weight PEO (Mv ≈ 600,000 g mol?1) matrix. The PD copolymer could easily dissociates into PEG-PDMAEMAH+ (PD+) polycations and high-donor-number NO3? anions. The large, flexible PD+ cation provides excellent solubility for NO3?, overcoming the solubility limitations of traditional nitrate additives. Simultaneously, at the macro/mesoscopic level, the good compatibility between PD+ and PEO effectively promotes PEO amorphization and chain segment motion, avoiding the phase separation and ion blocking issues associated with conventional additives or fillers.
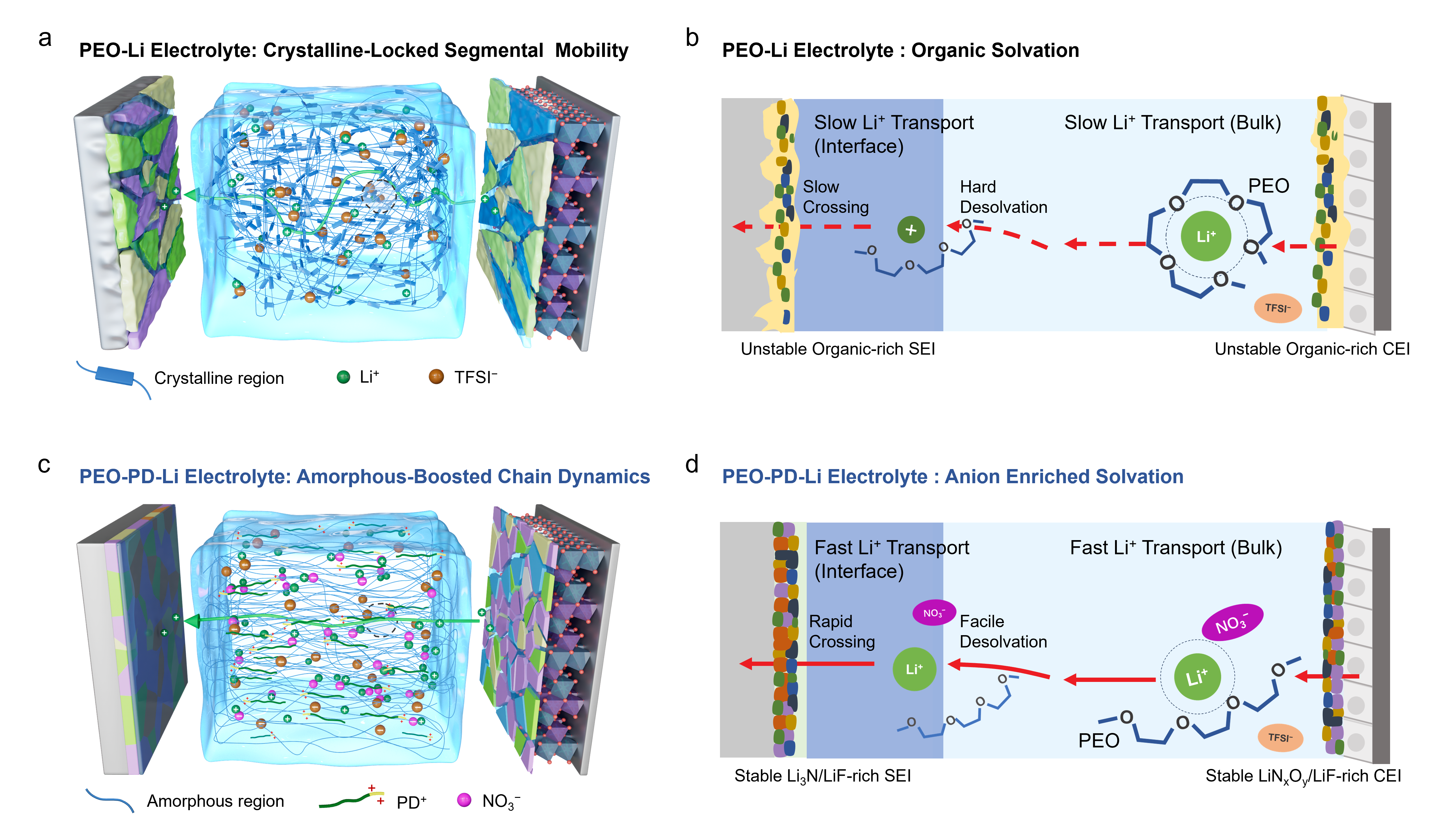
Figure 1 Schematic diagram of the proposed multiscale engineering strategy for PEO electrolytes.
At the microscopic level, NO3? anions, possessing a high donor number (22.2 kcal mol?1), preferentially compete for Li+ coordination, weakening the strong chelation between EO and Li+, thereby enhancing bulk Li+ mobility. This simultaneously fosters an anion-rich solvation structure. This structure not only lowers the Li+ desolvation energy barrier but also facilitates the formation of robust, highly ion-conductive CEI (rich in LiNxOy/LiF) and SEI (rich in Li3N/LiF) layers at the cathode and anode interfaces, respectively, enhancing interfacial kinetics and high-voltage tolerance. Experiments demonstrated that the PD-modified PEO electrolyte enables solid-state lithium metal batteries to operate stably near room temperature (30 °C) without any liquid plasticizers. Consequently, high-voltage LiNi0.8Co0.1Mn0.1O2 (NCM811) cells achieved over 500 stable cycles at a 0.2 C rate with a high capacity retention of 82.7%, while LiFePO4 cells operated stably for over 1200 cycles at 0.5 C.

Figure 2 Microscopic solvation regulation.

Figure 3 Interfacial property characterization.
This research simultaneously achieves three major goals for PEO-based all-solid-state batteries of room-temperature operation, high-voltage compatibility, and long-term stability, through multiscale synergistic regulation. It systematically reveals the critical role of anion chemistry in polymer electrolytes. This molecular design strategy opens a feasible pathway towards the commercial application of solid-state batteries.
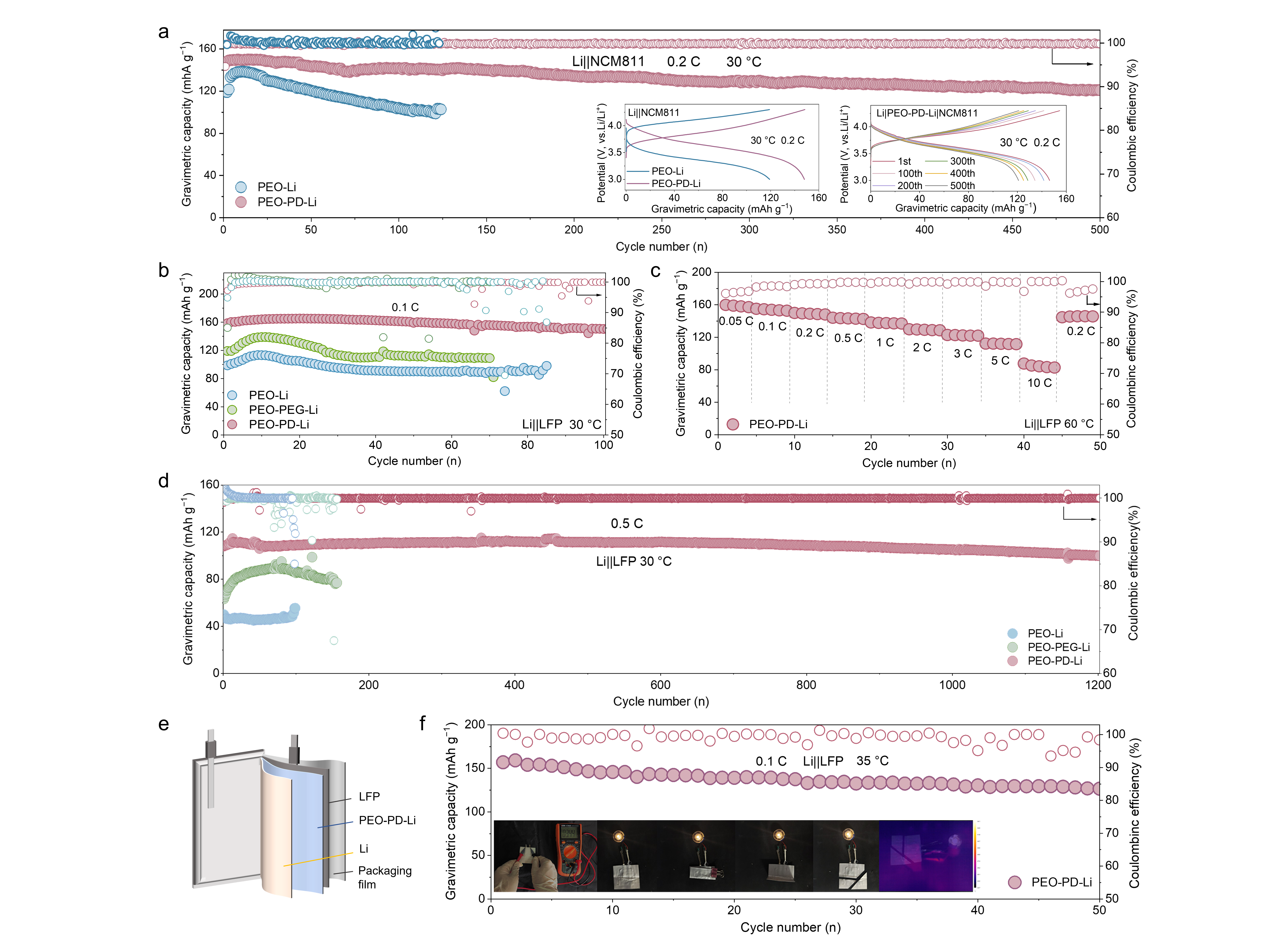
Figure 4 Electrochemical performance.
The research was supported by the National Natural Science Foundation of China, the Natural Science Foundation of Shandong Province, the China Postdoctoral Science Foundation, and the Youth Innovation Talent Introduction and Education Program for Universities in Shandong Province.
Link to the full paper: https://doi.org/10.1002/anie.202523382

